

The Minuteman® technology consists of thoughtfully-designed instrumentation and implants that enable providers to perform all of the necessary elements to achieve fusion of the posterior bony elements of the lumbar spine.
The tissue-sparing approach uses a series of instruments to access and enable direct visualization of the posterior bony elements (e.g., posterior bony anatomy, laminae, facet joints, etc.) and to decorticate bony surfaces using two specialized decortication instruments. Next, bone graft materials are placed in the implant and delivered directly in contact with the freshly decorticated bone. The implant is placed under compression between the bony surfaces to encourage osseointegration and is then locked in place to span the interspace with secure fixation points at each end of the construct.
By strictly adhering to known principles of lumbar arthrodesis, Skoblar et al. recently reported successful fusion outcomes in 93% of patients utilizing the Minuteman® technology.
U.S. FDA Cleared Indications for Use
The Minuteman® G5 Fusion Plate is a posterior, non-pedicle fusion device, intended for use at a single interspace in the non-cervical spine (T1-S1). It is intended for plate fixation/attachment to the posterior bony anatomy for the purpose of achieving instrumented posterior arthrodesis (i.e., fusion) in the following conditions:
- Lumbar Spinal Stenosis;
- Degenerative Disc Disease (DOD) (defined as back pain of discogenic origin with degeneration of the disc confirmed by history and radiographic studies); and/or
- Spondylolisthesis.
The Minuteman® GS MIS Fusion Plate is intended for use with bone graft material. The device may be implanted via a lateral transverse approach (L1-S1) or a posterior approach (T1-S1).
Features & Benefits
Lateral Approach
______
Ligament Preservation
______
Of the supraspinous ligament helps maintain stabilization of the spine
Hydroxyapatite Coated Fixation Hex Nut
______
A substance that helps promote bone on-growth and fusion. Maximizes bone-implant contact area to promote stability and fusion.
Large Graft Window
______
MR Conditional
______
The Minuteman® G5 implant is considered MR Conditional. A patient may be safely scanned under very specific conditions, which may be found by visiting the MRI Safety.
5 Anatomical Sizes
______
(8mm, 10mm, 12mm, 14mm, and 16mm) to accommodate variations in patient anatomy.
Threaded Body
______
Sterile Packed
______
Promotes patient safety and compliance at hospitals

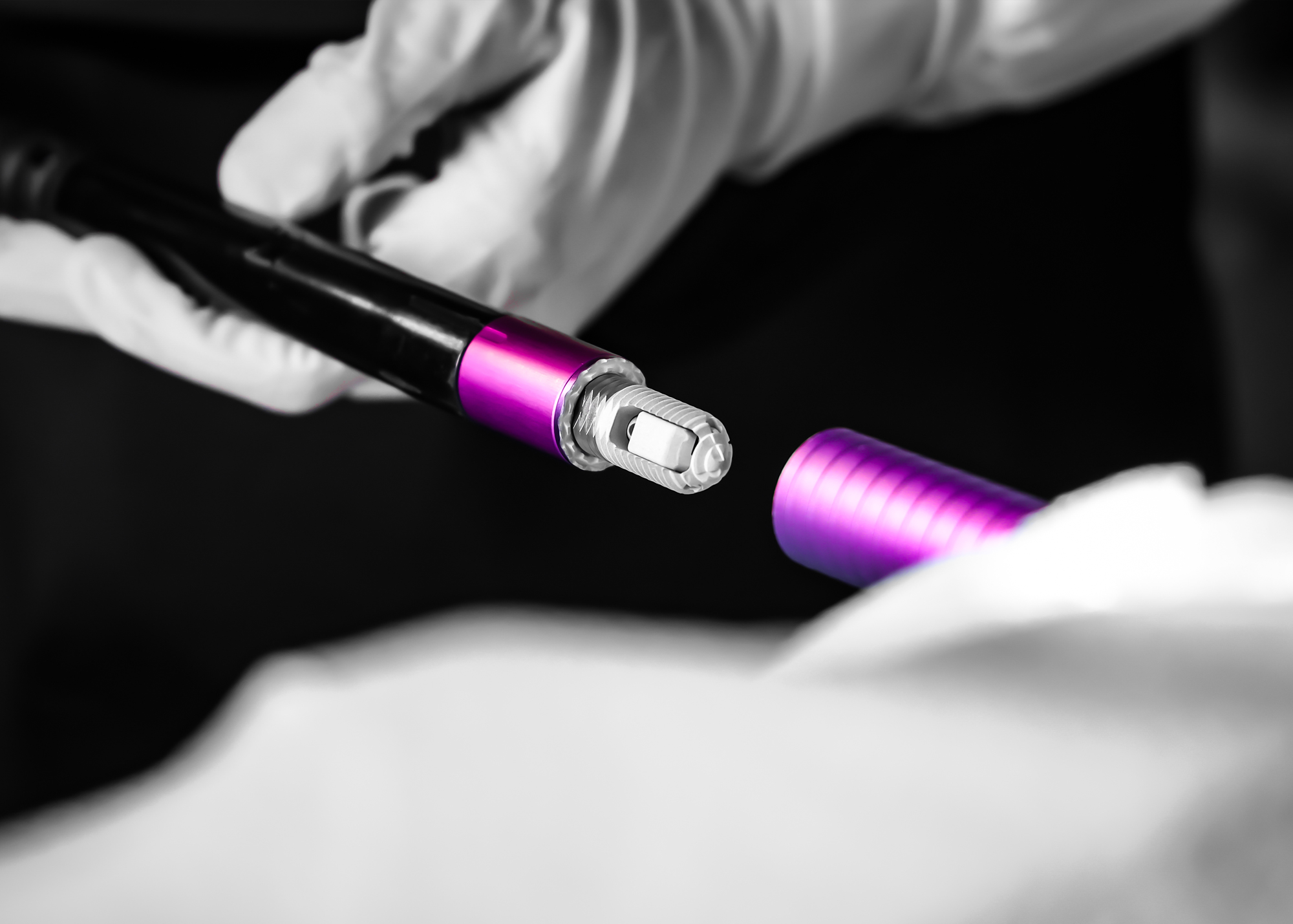

Single-Use Instrumentation Set
Our Single-Use Minuteman® Instrumentation Set exemplifies elegance and precision in medical technology, meticulously crafted to meet the highest standards of interventional pain management. This innovative set is designed to be ready for surgery right out of the box, eliminating the need for sterilization and significantly reducing preparation time.
Why Single Use?
-
Elegant Design: Sleek, user-friendly instruments that ensure ease of handling and optimal performance.
-
Precision Engineering: Each instrument is crafted with meticulous attention to detail, constructed for precise and reliable results.
-
No Sterilization Required: Designed for single-use, these instruments eliminate the need for post-procedure sterilization, saving valuable time and resources.
-
Efficiency in High-Volume Settings: The ability to use multiple sets in rapid succession enhances efficiency, allowing practitioners to maintain high standards of care even in busy surgical environments
-
The Minuteman® G5 Fusion Plate is intended for use with bone graft material. The device may be implanted via a lateral transverse approach (L1-S1) or a posterior approach (T1-S1).
Included in the Set:
-
Guidewire and Extension
-
Sequential Dilators and Working Sleeves
-
Facet Rasp and Graduated Tap
-
Minuteman Inserter
Experience the future of interventional pain management with the Single-Use Minuteman Instrumentation Set—where simplicity meets innovation leading to optimal patient outcomes.

What are Minuteman® Patients Saying?
Patients of Dr. Rick Pacius (Newport Beach, CA)
- “I had searing sciatica pain so bad I couldn’t hold my granddaughter. After Minuteman, the pain was gone.” — Dr. Pacius’ patient
- “I went in with pain and walked out pain-free the same day.” — Dr. Pacius’ patient
- “Now I lift weights, take long walks, and play with my granddaughter without worrying about my back.” — Dr. Pacius’ patient
Patients of Dr. Chris Bovinet (Brunswick, GA)
- “Minuteman relieved my leg pain immediately and gave me my life back.” — Dr. Bovinet’s patient
